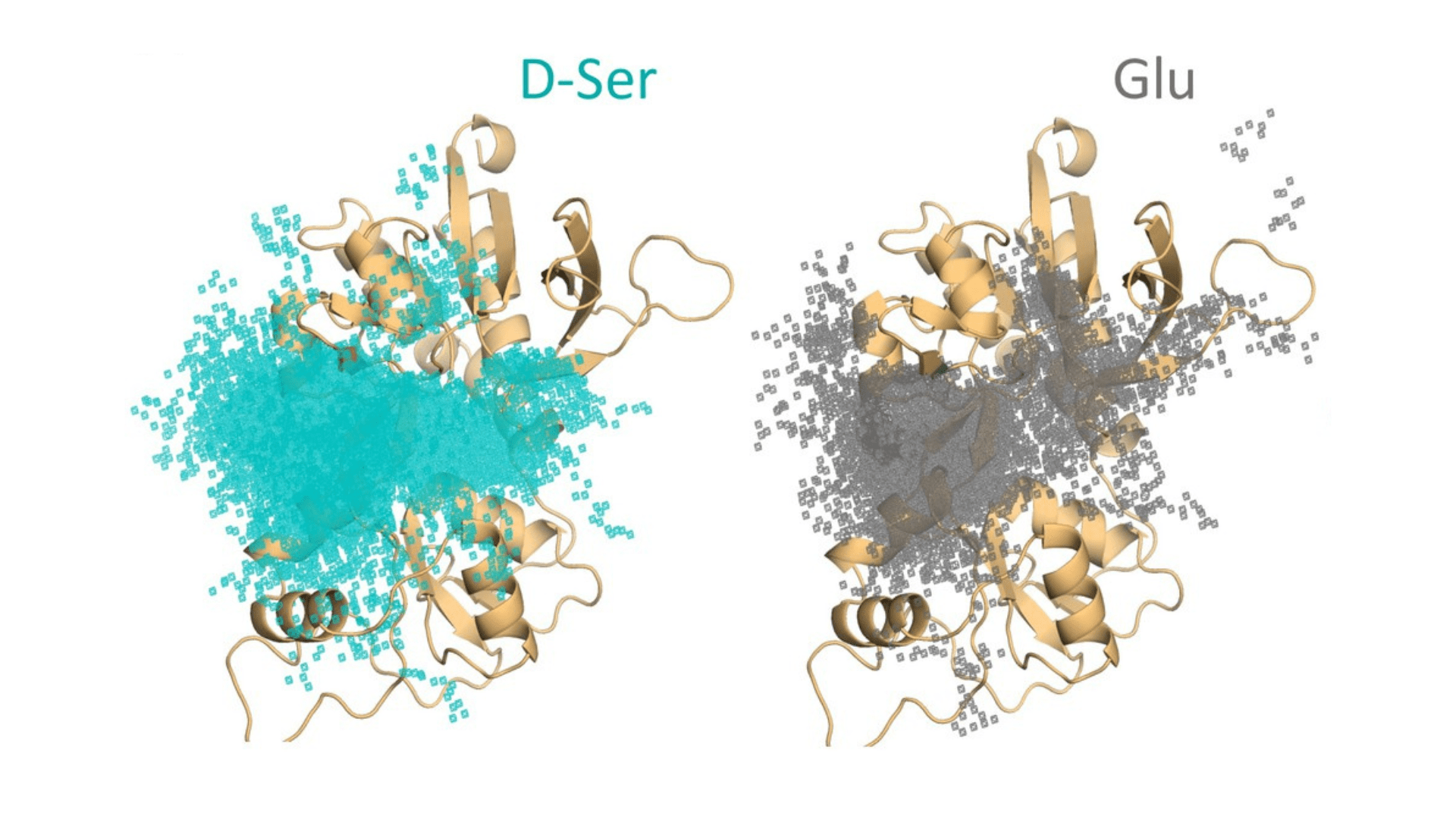
Comparison of where D-serine (D-Ser, teal dots) and glutamate (Glu, gray dots) stick to the GluN2AA subunit. From Remy A Yovanno Tsung Han Chou Sarah J Brantley Hiro Furukawa Albert Y Lau (2022) Excitatory and inhibitory D-serine binding to the NMDA receptor eLife 11:e77645.
Ability of D-serine to switch from boosting excitatory signals to blocking them suggests route for preventing long-term injury or stroke damage
D-serine is important for strengthening signals from glutamate, the brain’s chief neurotransmitter for exciting is nerve cells. But a team using the Anton 2 supercomputer hosted at PSC to simulate D-serine interacting with a key brain cell-surface protein found that, at high enough levels, D-serine can change from strengthening glutamate signals to blocking them. The unexpected discovery, confirmed by lab experiments, opens the possibility of future treatments to halt the nerve cell overactivity that causes lasting disability after brain injury or stroke.
WHY IT’S IMPORTANT
The working of our brains depends on a complicated dance of signals between nerve cells. Nerve cells can excite each other into higher activity or inhibit each other from firing. It’s the pattern of which signals are transmitted and which are blocked that forms our memories, our thoughts, our emotions.
Central to this pattern are the excitatory neurotransmitters. These small molecules are the messengers that cross the tiny gap between a signaling nerve cell and its target cell, called the synaptic cleft. Medically, these neurotransmitters are important because their over-activity in the brain in the wake of traumatic brain injury or stroke can kill neurons and cause lasting disability.
The chief excitatory neurotransmitter in vertebrates is the amino acid glutamate. One of the main families of cell-surface receptor proteins that responds to glutamate is the N-methyl-D-aspartate (NMDA) receptor.
In the NMDA receptor, glutamate doesn’t act alone. Two of the four protein subunits that make up the NMDA receptor, called the GluN2A subunits, dock with glutamate. The other two subunits, called GluN1, dock with either of the amino acids D-serine or glycine. The docking of glutamate together with either D-serine or glycine is necessary for starting an excitatory signal within the cell.
“The NMDA receptor is involved in mediating communication between neurons. When one neuron wants to communicate with another, it releases glutamate into the synaptic cleft. Some glycine or D-serine is there, too … There are biological consequences; over-activation of NMDA receptors is involved in neuronal damage resulting from stroke.” — Albert Lau, Johns Hopkins University School of Medicine
Albert Lau and his team at Johns Hopkins University School of Medicine wanted to understand the differences in docking processes between glutamate and D-serine to the NMDA receptor. They realized that, to do that, they’d need to carry out long-timescale computer simulations of the movement of the receptor when it encounters both transmitters. To perform this massive computation, they chose Anton, a special-purpose supercomputer for molecular dynamics (MD) simulation that was designed and constructed by D. E. Shaw Research (DESRES).
HOW PSC HELPED
Graduate student Remy Yovanno, working in Lau’s lab, used Anton 2 to carry out a series of MD simulations, using the known properties of the NMDA receptor and its protein chains to see how binding to D-serine and glutamate changed their movements. A second-generation Anton machine, made available without cost by DESRES, is hosted at PSC, with operational funding support by the National Institutes of Health.
Important for getting to the bottom of D-serine’s activity would be carrying out the MD simulations for as much simulated time as possible. This was because the scientists didn’t know how the molecule would nestle into the NMDA receptor’s structure. Previous studies had shown that transmitters can enter a receptor’s binding site via different paths. They can also wind up in unexpected places in the protein. To fully sample the behavior, Yovanno would need to carry out the simulations for as long as she could.
Anton, which DESRES developed to vastly accelerate the process of MD simulation, was ideal for the work. It has a unique ability to carry out long simulations that are not possible in a reasonable amount of real time using general-purpose supercomputers.
“Anton was crucial [for this work]. Initially, when we approached this project, we wanted to look at differences in binding pathways to the NMDA receptor between glutamate and D-serine. Anton 2 was the perfect resource to use for this computational experiment because it required long-timescale simulations … to look at these different binding pathways.” — Albert Lau, Johns Hopkins University School of Medicine
Yovanno simulated D-serine docking with a kind of stripped-down version of the NMDA receptor to simplify the computations. She found something nobody expected.
While D-serine could and did stick to the GluN1 subunits of the receptor, it also sometimes bound to the GluN2A subunits. Scientists hadn’t realized this was possible. Yovanno investigated which parts of the GluN2A protein structure were responsible for this interaction, identifying key parts of the receptor’s surface that interacted with D-serine.
The discovery wasn’t just surprising. It also opened an intriguing possibility. If D-serine could bind to GluN2A, was it possible that it could block glutamate from binding to the protein? Such interference happens in living cells. It’s often a key means for controlling signals.
To investigate this question further, and to get a crucial reality check on the simulations, Lau’s team turned to colleagues at Cold Spring Harbor Laboratory. Hiro Furukawa’s lab there had performed fundamental experiments with the NMDA receptor. They could take on the job of testing this new activity of D-serine in live cells.
Furukawa’s group found that the computer simulations were right. If they added enough D-serine, the transmitter changed from increasing glutamate’s signal to interfering with it. The collaborators reported their results in the journal eLife in October 2022.
The scientists continue to work with the NMDA receptor system and the transmitters that activate it. A key question is whether D-serine levels can become high enough in living brains to start blocking glutamate, as opposed to an effect that only happens in the lab. Another intriguing possibility is that a drug based on D-serine or a molecule that mimics it could possibly block overactive glutamate signaling in an injured brain, preventing long-term disability following injury or stroke. It’s only an idea now, and hasn’t yet been tested in the lab, let alone in human patients. But the scientists believe it’s worth pursuing.
