A two-step method identifies known drugs likely to reduce chance of death from covid-19
Bridges helps identify meds for other conditions that associate with lower death rates in COVID-19 patients
by Ken Chiacchia
Both the ongoing treatment of COVID-19 and readiness for the next pandemic or other health crisis require faster development of new medications. The startup company ARIScience is developing a computational platform to repurpose drugs to new diseases. They used PSC’s Bridges system in a two-step process. First they used high performance computing (HPC) to simulate known drugs’ ability to interfere with proteins in the virus. Then, using non-HPC systems, they identified which of the most effective of these drugs associate with lower death rates in patients with COVID-19 taking the medications for other conditions.
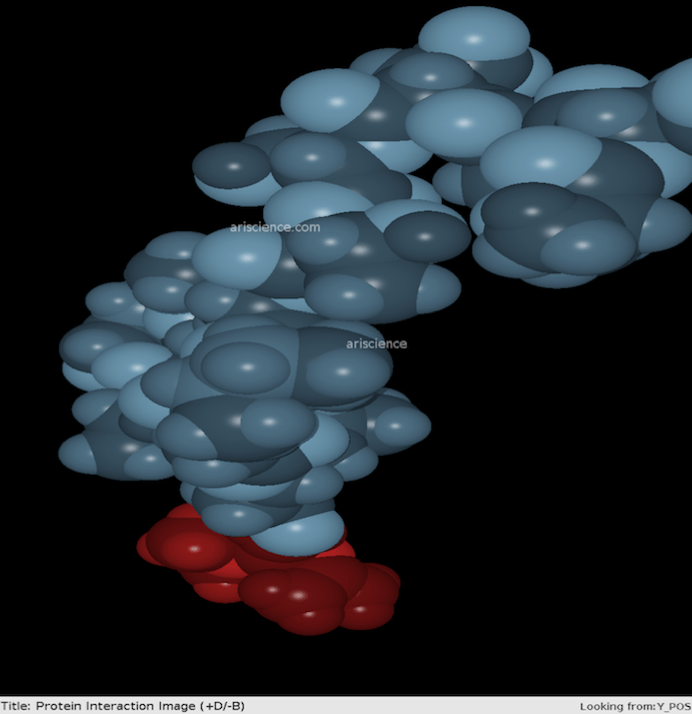
Simulated image of metformin (red), interacting with SARS-CoV-2 protein (blue) at neutral pH.
Why It’s Important
Among the many lessons of COVID-19 is that medical research sometimes needs to be fast as well as good. The rapid development of SARS-CoV-2 vaccines was a scientific triumph, but it didn’t really happen in less than a year—scientists had been working on vaccines for the SARS-CoV virus family since it was discovered in 2003. Plus, the next health crisis is a “when,” not an “if.” It may require an even more rapid response from a much colder start. In addition, the ongoing race between vaccination and the spread of the SARS-CoV-2 virus (including new variants that might eventually evade the effectiveness of the current vaccines) means we still need new drugs to combat it. We need to figure out what we can do in a short period with “off the shelf” tools when a new health threat emerges.
“When COVID-19 came to the US, we’d already been working on our novel simulation and repurposing platform for about two years. So this is something we’d been creating not because of COVID-19, but because our objective was to do drug repurposing—using existing compounds … pretty much any compound that has passed phase 1 [FDA-mandated safety testing].”—Joy Alamgir, ARIScience
Scientist Joy Alamgir founded a Massachusetts-based biotech startup company, ARIScience, to see whether he could screen the drugs we have today, already known to be safe, for activity against disease pathways. His idea was simple. If he could find existing drugs that in a computer simulation interrupt key proteins in a disease pathway, and then looked at a large enough, de-identified and detailed patient dataset, would those drugs show a measurable difference in patient outcomes?
Alamgir did this without knowing at the time that the COVID-19 pandemic was coming. The pandemic became a potentially lifesaving testing ground for his company’s ideas. Using PSC’s Bridges platform and other HPC centers across the country, his team swung into action, eventually with an allocation via the COVID-19 HPC Consortium. They used this computing time to search for the drugs most likely to interfere with replication of the SARS-CoV-2 virus in host cells. The team included scientists at ARIScience, Boston University, the Great Plains Tribal Leader’s Health Board, Caromont Regional Medical Center, the National Institutes of Health (NIH), Oregon Health & Science University, Johns Hopkins University and Brown University.
How PSC Helped
To start out, the team looked at 11 proteins from SARS-CoV-2 and 1,513 FDA-approved drugs. They used HPC simulations to see which of the drugs are likely to stick to and interfere with critical parts of the proteins. Using non-HPC computing, they then took the 18 drugs with the most potential effectiveness and searched the National COVID Cohort Collaborative (N3C) database for patients taking those drugs. Using 1.5 million anonymized COVID-19 patients, they then set-up matched cohorts–comparing patients with similar conditions–to compare mortality odds in patients taking and not taking these 18 medications. They used 18 predictors to match patients, including body mass index, other disease processes, and prior access to healthcare. They performed the cohort matching to isolate effects that were likely to be from the drug of interest and not other factors.
ARIScience had a small HPC cluster available, but this task was well beyond its capacity. To meet this need, they used a number of HPC systems. PSC’s National Science Foundation-funded Bridges platform in particular offered the ability to conduct many simulations of a given protein at once allowing simultaneous testing of the drugs against vulnerable points on each of the 11 proteins. Alamgir’s team also used time on the Stampede2 supercomputer at the Texas Advanced Computing Center, a member with PSC in the NSF’s XSEDE network of advanced research computing centers.
“Bridges—particularly its regular-memory partition—was very useful to us … We took the proteins of interest, virtually deconstructed it, and figured out areas that we should be able to target. For each protein we then simulated against the 1,513 drugs in our library. For each protein that we simulated on Bridges we created 800 jobs for Bridges to allocate across whatever nodes were available.”—Joy Alamgir, ARIScience
The simulations produced a ranking of the 1,513 drugs, identifying 18 with the most potential activity against specific proteins of SARS-CoV-2. When the team compared mortality odds among matched COVID-19 patients who were and weren’t taking these 18 drugs, four drugs emerged as the most effective. The diabetes drug metformin, the glucocorticoid/skin medicine triamcinolone, the antibiotic amoxicillin and the diuretic/first-line-blood-pressure medication hydrochlorothiazide reduced the odds of mortality between 23 and 27 percent in their analysis. Their work is now in submission with a scientific journal.
The researchers’ task isn’t over for identifying COVID-19 drugs. One important question is why some of the drugs identified in ARIScience’s study had not shown an effect against the virus in previously testing. Alamgir thinks that the tests used for these drugs likely stressed stopping the virus’s ability to kill host cells, rather than a more subtle effect of delaying viral activity. That distinction is key, as delaying the course of infection gives the patient’s body more time to mount an effective immune response. Still, the four “winning” drugs are promising, and represent a major proof of concept for ARIScience’s approach in drug searches for future health problems. Another goal for ARIScience is autonomous de-novo drug construction, using their current computational platform for the next step of automated design of new drugs with a desired clinical activity.

