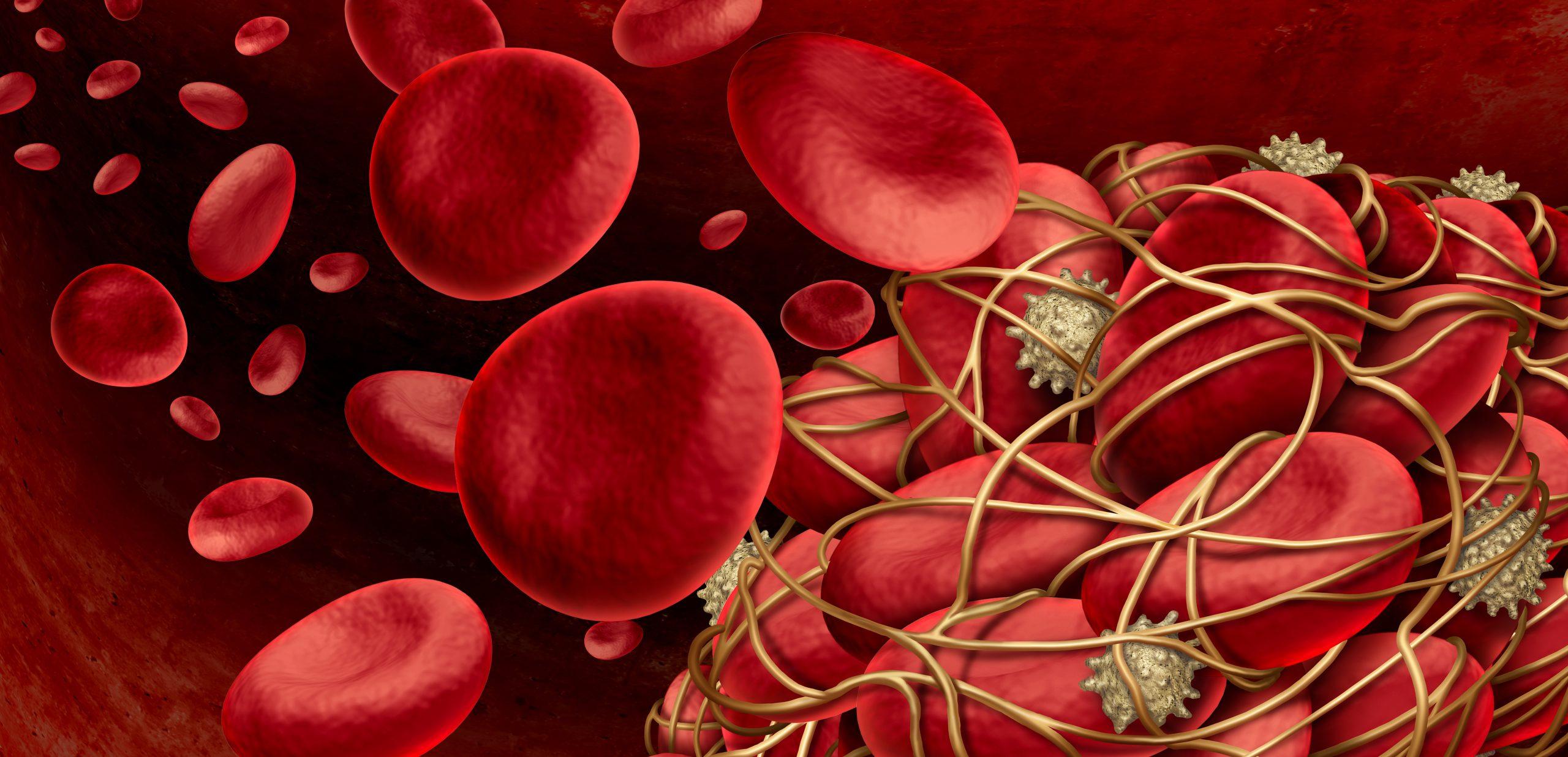
Developing blood clot, with fibrin “cables” (yellow), being pulled together by platelets (off-white). Clots that trap more red blood cells (red) may not contract as effectively, slowing healing. Adobe Stock image: 240_F_453411695_2N2Jddctb1vWwqowio9hVtKoNrZ76X65.jpg
Addition Is Latest Step in Building First-Principle Model of How Clots Form and Function in Health and Disease
WHY IT’S IMPORTANT
The phenomenon of blood clotting is huge in human health. We need it to recover from the most minor wounds. When it isn’t needed, it can lead to life-threatening conditions like heart attack, stroke, and pulmonary embolism in the lungs. When it doesn’t happen quickly enough, it underlies hemophilia and other bleeding disorders.
Of course, people have known that blood clotting leads to healing since ancient times. In 1835, Andrew Buchanan in Scotland proposed there was a factor in blood that caused it to happen. In 1882, Giulio Bizzozero discovered platelets and the fact that they clump together to begin the clotting process. 1905 saw Paul Morawitz identifying the four components needed to start the clotting cascade.
“Blood clotting is a very important process … essentially it’s the way for us to repair ourselves. When we get injured, the process goes on to restore blood circulation. On the other hand, it is a delicate process that, if it works too intensely … you can have a stroke or other problems. When it doesn’t work, you can bleed and bleed.” — Alexander Alexeev, Georgia Tech
Diana Sun, then a graduate student in Professor Alexander Alexeev’s laboratory at Georgia Tech, wanted to understand whether blood clot formation could be simulated by simplifying the problem by focusing on mechanical forces only. If the idea was correct, scientists could use a straightforward computer model to study the roles different components play in healthy and disease-associated clot formation. To recreate this phenomenon in a computer, the team turned to PSC’s NSF-funded Bridges-2 supercomputer.
HOW PSC HELPED
Blood clots begin to form when platelets stick to the tissues around a wound site. This forms a temporary plug in the wound. The platelets also release factors that, in combination with other proteins and chemicals in the blood, form a scaffold of fibrin protein. The platelets then use fibrin to pull the wound closed, like little winches pulling on cables. Once the wound is closed up enough, the network of fibrin cements the platelets together to form a stable clot.
Sun, who is now an assistant professor of mechanical engineering at Lafayette College, started with simple assumptions about the platelets and fibrin strands. Would this computer model act the same way that real clots do in the body?
Despite this simplification, the simulation would require a lot of computer power. In particular, it would need to take advantage of a method called large neighborhood search, finding “good-enough” solutions to the problem of how the platelets and fibrin would interact by sampling the components over a relatively large area. This approach takes a lot of computer memory, the same as RAM on a laptop. For some supercomputing systems this would be a “large-memory” problem. But Bridges-2 has 256 to 512 gigabytes of memory in its regular memory nodes — about 500 times what you’d find in a high-end laptop, but in each of hundreds of nodes. PSC’s flagship computer had no problem tackling the task. The scientists got time on Bridges-2 through an allocation from ACCESS, the NSF’s network of high performance computing resources.
“Bridges-2 is very essential for our work … With [it] I have sufficient memory in the nodes — memory that my advisor’s cluster couldn’t handle.” — Diana Sun, Lafayette College
Sun carried out a series of simulations on Bridges-2. At first these simulations involved the minimum number of factors needed to make a clot form. With time, she added more and more factors. In the most recent simulation, she added the RBCs. At every step, the team turned to laboratory experiments with real blood clot formation to confirm that the simulated clots were behaving the same way as real ones.
In a paper in the Biophysical Journal in March 2023, the scientists reported that RBCs seem to collect in blood clots just because the contracting fibrin mesh traps them there. Interestingly, the more RBCs trapped in the clot, the harder it is for the platelets to pull the wound closed. When some RBCs at the outer margins of the clot escaped, the simulated clot formed a dense fibrin outer shell that looked a lot like thrombus clots seen in the real world. These clots underlie conditions like deep vein thrombosis, which can cause pain and swelling in the legs.
To date, the simulations only deal with how static clots form and how different factors like RBCs affect the process. In current and future work, Alexeev and his collaborators are investigating what, if any, role the phenomena in the virtual clots play in real disease processes. A big next step will be to add the flow of blood past and through the clot to the simulation. Another goal will be to find out how these phenomena affect the penetration of different types of clot-busting drugs to help the treatment of heart attacks and strokes.
